My name is Sanne Derks and I am a medical device specialist focused on delivering high quality, no nonsense clinical services.
In need of some common sense? Give me a call or send me an e-mail.
More about meDicuss your situation, ask specific questions or take a look at our quality system.
Click hereOn 26 May 2021, EU regulation 2017/745 (Medical Device Regulation, MDR) is scheduled to come into full effect, thus amending the Medical Device Directive (MDD).
More about Medical Device Regulation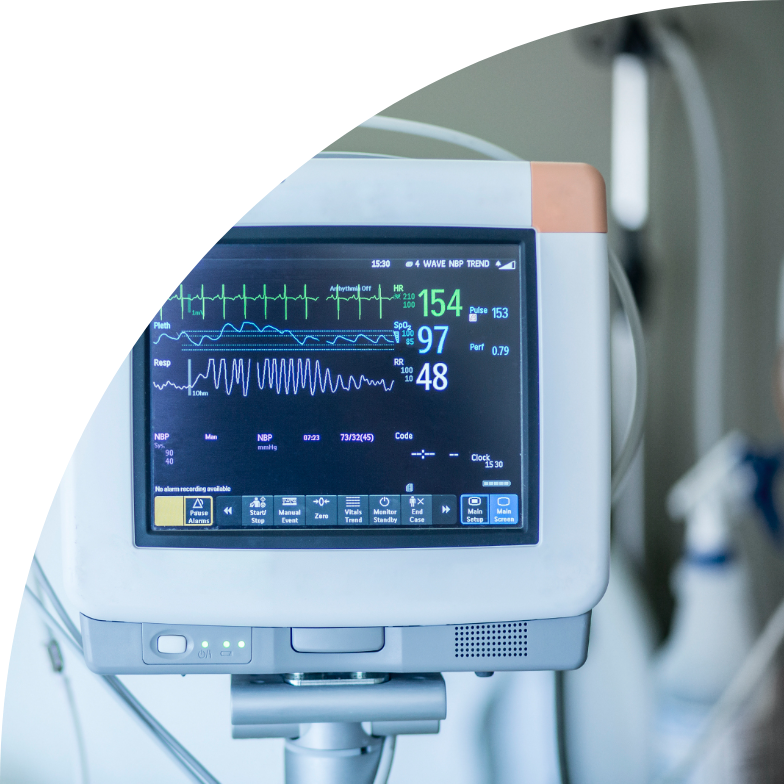
A well-designed clinical investigation needs a well-designed clinical Quality Management System (QMS) as a firm basis to effectively protect subject rights, safety and wellbeing, and ensure high quality data. Additionally, a good QMS will provide maximum efficiency and flow throughout your clinical investigation instead of compromising your progress.
More about Clinical Quality Management System



My name is Sanne Derks and I am a medical device specialist focused on delivering high quality, no nonsense clinical services.
In need of some common sense? Give me a call or send me an e-mail.
More about me